This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
GDF15 hormone found to make insulin work better
More than 500 million people worldwide suffer from diabetes. Roughly 2.6 billion are overweight. Something that both groups have in common is that the hormone insulin does not work nearly as well for them as it does for people who are healthy and in a normal weight range. Researchers have been eager to better understand the processes that affect insulin function in our bodies in order to develop new medications and treatments.
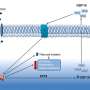
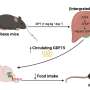
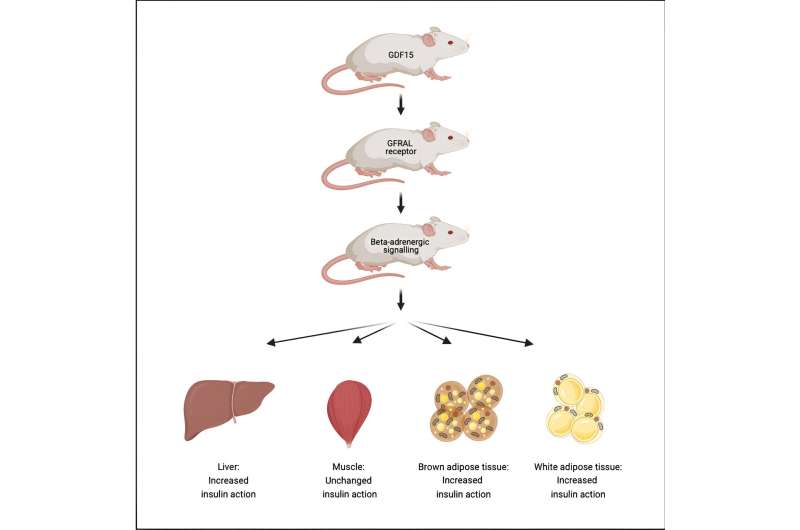
In a new study, researchers from the University of Copenhagen's Department of Nutrition, Exercise and Sports (NEXS) studied how GDF15 (growth differentiation factor 15) affects insulin sensitivity in rodents.
The researchers were the first to discover that GDF15 actually improves sensitivity in mice and rats, making them better suited to regulate their blood sugar and absorb energy in their muscles. The result, published in journal Cell Metabolism, increases our general understanding of GDF15.
"Increased insulin sensitivity is an important indicator of health and critical for avoiding type 2 diabetes, high blood pressure and cardiovascular diseases. That we are now able to demonstrate a beneficial effect of GDF15 in rodents raises interesting perspectives," explains Professor Erik Richter, the study's senior author.
Obese rats lose weight, lean ones benefit from improved insulin action
Among other things, previous studies demonstrated that the hormone caused obese rodents to lose weight because the hormone acts as an appetite suppressant.
In this study, the researchers wanted to see if lean rodents benefited from receiving the GDF15 hormone, even though they did not lose weight. The experiments showed that a low dose of GDF15 every other day, spread over three occasions, improved insulin action.
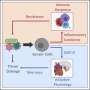
"Because quite high doses of GDF15 are needed to curb appetite, our lean rodents didn't lose weight with the low doses that we gave them. However, their insulin sensitivity improved. It turned out that it was primarily their liver and fatty tissue that increased insulin sensitivity, and not the muscles, as we had anticipated," says Richter.
High insulin sensitivity is a good health indicator because lowered insulin sensitivity—called insulin resistance—strains insulin secretion in the pancreas and can lead to the onset of type 2 diabetes, among other things.
The hormone's ability to act as an appetite suppressant has also been tested in humans—but without quite as positive results, as nausea was a side effect. However, nausea is also seen with other appetite suppressant formulations. Whether GDF15 also increases insulin sensitivity in humans remains to be studied.
Many strange effects in the body
The GDF15 stress hormone is not unknown among scientists due to it being secreted in a very diverse range of physiological states. For example, GDF15 concentrations increase during physical activity, with aging, and in people who are overweight or battling serious diseases like cancer or heart disease. In pregnant women, levels of the hormone are elevated 100 times above normal. The better a person's physical fitness, the lower their GDF15 levels are in the bloodstream while at rest.
One could be tempted to consider GDF15 as an inverse biomarker of general health—the lower the GDF15 while at rest, the better. But as with all of the other physiological conditions in which this mysterious hormone is in some way involved, the researchers still do not know exactly what role GDF15 plays:
"The hormone stands out because it is secreted in so many different situations without us fully understanding why and what its effect is. On one hand, we can see that it improves insulin sensitivity in mice, which is a positive physiological effect. But at the same time, it's a hormone that increases under various types of stress. How exactly this is all connected is something that we need to investigate more thoroughly," concludes Erik Richter.
More information: Kim A. Sjøberg et al, GDF15 increases insulin action in the liver and adipose tissue via a β-adrenergic receptor-mediated mechanism, Cell Metabolism (2023). DOI: 10.1016/j.cmet.2023.06.016