Cells too stiff to scavenge leads to lupus, an autoimmune disease

More than 50 billion cells die in the human body every day, a spectacle of programmed cell death called apoptosis. These cells undergo internal degradation and then fracture into apoptotic bodies that are scavenged by immune cells, all without triggering the body's immune system defense.
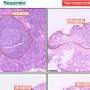
One of the most dangerous places for cells to die is near the follicles of the spleen. The follicles are primed to mount intense immune attacks against infectious bacteria or viruses. If an apoptotic cell (AC, a cell undergoing programmed cell death) goes into the follicle, it can trigger an autoimmune response. This can lead to autoimmune diseases like systemic lupus erythematosus, where the immune system attacks its own body.
Macrophages in the spleen marginal zone around the follicles keep the ACs out, acting like the defensive line in football or a "final exclusion barrier," says John Mountz, M.D., Ph.D., professor of medicine in the University of Alabama at BirminghamDivision of Clinical Immunology and Rheumatology.
In a paper published online in the Journal of Clinical Investigation on June 22, Mountz and colleagues, including Hui-Chen Hsu, Ph.D., and Hao Li, Ph.D., present a new model of why this defensive line becomes porous in mouse models of lupus, and they show the mechanism that underlies this breakdown. Just as "location, location, location" is the difference between good and bad real estate, the Mountz group and colleagues say that the location of immune system B cells is a key difference between healthy animals and those that develop autoimmune disease. The wrong location leads to macrophage cells that have problems in their cytoskeletons—they become too stiff to engulf and digest dead cells.
Mountz and colleagues found that the presence of B cells in the marginal zone outside the follicle is essential to maintain the defensive line of macrophages. They found that type I interferon—which is elevated in lupus—causes the B cells to migrate away from the marginal zone and into the follicle, which interrupts vital cross-talk between the B cells and the marginal zone macrophages. In this model, the definition of an autoimmune pathogenic B cell is now based on its anatomic location.
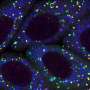
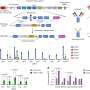
Experiments by the Mountz group show the mechanism of this cross-talk: B cells in the marginal zone of the follicle interact with the marginal zone macrophages by means of a membrane lymphotoxin present on the surface of the B cells. This lymphotoxin binds to a lymphotoxin receptor on the surface of the macrophages. That connection stimulates a mechanosensing complex in the macrophage, triggering the production of a gene regulator called megakaryoblastic leukemia 1 (MKL1) that regulates the cell's actin cytoskeleton and enables a macrophage to respond to and engulf ACs. Without that B cell interaction, MKL1 expression in the macrophage decreases, changing the activity of the actin cytoskeleton.
This signaling axis—from B cell to macrophage to mechanosensing signaling—keeps the defensive line of macrophages strong and healthy. In contrast, mice that have had the MKL1 gene knocked out begin to resemble lupus-model mice as they age. They show decreased AC clearance, a deficiency of macrophages in the marginal zone of spleen follicles and increased production of autoimmune antibodies. This suggests a key role for macrophage mechanosensing signaling in lupus.
Examination of slides of spleens from human systemic lupus erythematosus patients showed a pattern similar to that seen in two mouse models of lupus: reduced numbers of B cells surrounding the follicle, increased numbers of B cells inside the follicle and a loss of MKL1 expression in the marginal zone.
"A cell doesn't do anything unless it senses its environment," said Mountz, whether the sensation is heat, chemical signals, oxygen availability, mechanical touching or other stimuli. "We knew that, in lupus, there was an overwhelming production of antibodies against materials derived from ACs. Researchers thought that this may be related to failure of macrophages to eat and digest ACs. Many possibilities have been suggested, but no one knew the basic cause."
"This paper shows that the immune system is also regulated by mechanosensing," Mountz said.
He says this paper is the first connection between poor agility of a cell (the movement of the macrophage and its engulfment of ACs, as a result of chronic type I interferon stimulation) and the etiology of lupus.
The paper is accompanied by a commentary in JCI by Claudia Mauri and Madhvi Menon, University College London, titled, "The many faces of type I interferon in systemic lupus erythematosus." They write that, "The results of this study provide important insight into factors that inhibit AC clearance and promote the development of systemic lupus erythematosus."
More information: "Interferon-induced mechanosensing defects impede apoptotic cell clearance in lupus." www.jci.org/articles/view/8105 … b4db007a335a44c9b4ac